Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Kato, Chiaki; Kiuchi, Kiyoshi; Sugimoto, Katsuhisa*
Corrosion Engineering, 52(1), p.69 - 85, 2003/01
It is necessary to know the generation mechanism of high equilibrium potential in the solutions. Existing nitrogen oxides in nitric acid solutions were first analyzed by Raman spectroscopy and then existing amount of nitrogen oxides were examined by thermodynamic calculation using the SOLGASMIX software. The Raman spectroscopic analysis showed that the existing amount of un-dissociated HNO increased with increasing nitric acid concentration and solution temperature. The thermodynamic calculation showed that the important nitrogen oxides in nitric acid solutions are HNO
increased with increasing nitric acid concentration and solution temperature. The thermodynamic calculation showed that the important nitrogen oxides in nitric acid solutions are HNO , NO
, NO
 , HNO
, HNO , NO
, NO , and NO. The equilibrium potential of nitric acid solutions is, however, mainly decided by the HNO
, and NO. The equilibrium potential of nitric acid solutions is, however, mainly decided by the HNO /HNO
/HNO equilibrium. The thermodynamic calculation also suggested that the increased oxidization potential on the heat-transfer surface is attributed to the reduction of nitrous acid concentration by the thermal decomposition of nitrous acid on the surface and the continuous removal of decomposition product from the solutions by boiling bubbles.
equilibrium. The thermodynamic calculation also suggested that the increased oxidization potential on the heat-transfer surface is attributed to the reduction of nitrous acid concentration by the thermal decomposition of nitrous acid on the surface and the continuous removal of decomposition product from the solutions by boiling bubbles.
Kato, Chiaki; Kiuchi, Kiyoshi; Sugimoto, Katsuhisa*
Zairyo To Kankyo, 52(1), p.44 - 52, 2003/01
In order to understand corrosion of metals in nitric acid solutions, it is necessary to know the generation mechanism of high equilibrium potential in the solutions, especially under boiling conditions. The Raman spectroscopic analysis showed that the existing amount of un-dissociated HNO increased with increasing nitric acid concentration and solution temperature. The existing amount of NO
increased with increasing nitric acid concentration and solution temperature. The existing amount of NO also increased by thermal decomposition. The thermodynamic calculation showed that the important nitrogen oxides in nitric acid solutions are HNO
also increased by thermal decomposition. The thermodynamic calculation showed that the important nitrogen oxides in nitric acid solutions are HNO , NO
, NO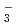 , HNO
, HNO , NO
, NO , and NO. The equilibrium potential of nitric acid solutions is, however, mainly decided by the HNO
, and NO. The equilibrium potential of nitric acid solutions is, however, mainly decided by the HNO /HNO
/HNO equilibrium. The thermodynamic calculation also suggested that the increased oxidization potential on the heat-transfer surface is attributed to the reduction of nitrous acid concentration by the thermal decomposition of nitrous acid on the surface and the continuous removal of decomposition product from the solutions by boiling bubbles.
equilibrium. The thermodynamic calculation also suggested that the increased oxidization potential on the heat-transfer surface is attributed to the reduction of nitrous acid concentration by the thermal decomposition of nitrous acid on the surface and the continuous removal of decomposition product from the solutions by boiling bubbles.
Sunaga, Hiromi
Denki Gakkai Gijutsu Hokoku, (895), p.61 - 62, 2002/09
no abstracts in English
Kojima, Takuji
Enerugi Rebyu, 22(4), p.27 - 29, 2002/04
no abstracts in English
Namba, Hideki
Hoshasen Riyo Ni Okeru Saikin No Shimpo, p.162 - 172, 2000/06
no abstracts in English
Hashimoto, Shoji
Denki Gakkai-Shi, 119(5), p.278 - 280, 1999/05
no abstracts in English
Tokunaga, Okihiro
Kankyo To Shoenerugi No Tameno Enerugi Shingijutsu Taikei, 0, p.506 - 508, 1996/00
no abstracts in English
Tokunaga, Okihiro
Genshiryoku Kogyo, 41(7), p.24 - 29, 1995/00
no abstracts in English
Tokunaga, Okihiro
Hoshasen To Sangyo, 0(66), p.59 - 60, 1995/00
no abstracts in English
*; Hirota, Koichi; *; *; *; *; *; *; *; Miyata, Teijiro; et al.
Radiation Physics and Chemistry, 45(6), p.1021 - 1027, 1995/00
Times Cited Count:7 Percentile:59.15(Chemistry, Physical)no abstracts in English
Tokunaga, Okihiro
Seidenki Gakkai-Shi, 19(4), p.296 - 300, 1995/00
no abstracts in English
Tokunaga, Okihiro; *; *
Hoshasen To Sangyo, 0(58), p.15 - 19, 1993/00
no abstracts in English
Namba, Hideki; Tokunaga, Okihiro; *; *; *; *
Radiation Physics and Chemistry, 42(4-6), p.669 - 672, 1993/00
Times Cited Count:15 Percentile:79.48(Chemistry, Physical)no abstracts in English
 , SO
, SO , and HCl removal from flue-gas ofmunicipal waste incinerator by electron beam irradiation
, and HCl removal from flue-gas ofmunicipal waste incinerator by electron beam irradiation*; *; *; Tokunaga, Okihiro; Miyata, Teijiro; Hirota, Koichi; *; *; *; *
Radiation Physics and Chemistry, 42(4-6), p.679 - 682, 1993/00
Times Cited Count:6 Percentile:55.97(Chemistry, Physical)no abstracts in English
Tokunaga, Okihiro
Isotope News, 0(450), p.8 - 11, 1991/12
no abstracts in English
Namba, Hideki; Tokunaga, Okihiro; *; *; *; *
Proceedings of the International Conference on Evolution in Beam Applications, 0, p.476 - 481, 1991/11
no abstracts in English
Namba, Hideki; Tokunaga, Okihiro; H.-R.Paur*
Journal of Aerosol Science, 22(1), p.475 - 478, 1991/00
no abstracts in English
;
Kogai To Taisaku, 22(6), p.537 - 543, 1986/00
no abstracts in English
 removal in electron beam treatment of exhaust gases
removal in electron beam treatment of exhaust gases; ;
Kogai To Taisaku, 19(2), p.158 - 164, 1983/00
no abstracts in English
 on the removals of NO and NO
on the removals of NO and NO in the moist mixture of O
in the moist mixture of O and N
and N
; ; ;
Int.J.Appl.Radiat.Isot., 30(1), p.19 - 23, 1979/00
no abstracts in English